Myelofibrosis causes
As with most blood cancers, scientists don’t know exactly why myelofibrosis happens. Sometimes it develops in people with other blood disorders, but most often, it arises in individuals without any known blood or bone marrow problems.
Hematopoietic stem cells, or blood stem cells, are the precursors to mature blood cells found mainly in the spongy center of bones, known as bone marrow. In myelofibrosis, these cells acquire genetic mutations that cause them to quickly produce large amounts of abnormal blood cells that don’t work properly. The chronic inflammation caused by these cancerous cells also leads to the buildup of scar tissue (fibrosis) that further inhibits healthy blood cell production.
While myelofibrosis can happen to anyone, there are certain risk factors that increase the chances it will develop. Among them are older age, radiation or chemical exposure, and pre-existing blood disorders.
Primary vs. secondary myelofibrosis
Myelofibrosis belongs to a group of blood cancers called myeloproliferative neoplasms (MPNs), in which the bone marrow overproduces blood cells. It is categorized into two types based on its origin:
- Primary myelofibrosis: This type develops spontaneously without another precursor condition and accounts for the majority of cases.
- Secondary myelofibrosis: This type develops secondarily to another condition, and accounts for as many as about one-third of cases.
Most often, secondary myelofibrosis occurs when other MPNs, namely essential thrombocythemia (ET) or polycythemia vera (PV), progress, and abnormal blood cell growth ultimately causes fibrosis. Less commonly, secondary myelofibrosis triggers could include other blood cancers, blood or bone marrow disorders, or immune conditions.
The role of gene mutations in myelofibrosis
In most people with myelofibrosis, cancer cells harbor disease-driving mutations in one of three genes: JAK2, CALR, or MPL. Mutations in these genes have also been linked to ET and PV.
These driver mutations are acquired, meaning they aren’t inherited, but rather develop spontaneously during a person’s lifetime. They’re also called somatic mutations.
Researchers don’t know why they happen, but they all lead to overactivation of the JAK-STAT signaling pathway that’s involved in cell growth and survival. When that pathway is consistently turned on, cancer cells continuously receive signals that allow them to grow uncontrollably.
As they multiply, these cells produce and release many different inflammatory molecules that further damage the bone marrow. Inflammation and bone marrow fibrosis impair healthy blood cell production.
In some people with myelofibrosis, mutations are not found in any of these three genes. This is known as triple-negative disease. It’s thought that in these cases, rare or unknown mutations may be involved.
The specific mutations found in a person’s cancer cells may affect blood cell production and the disease course in different ways:
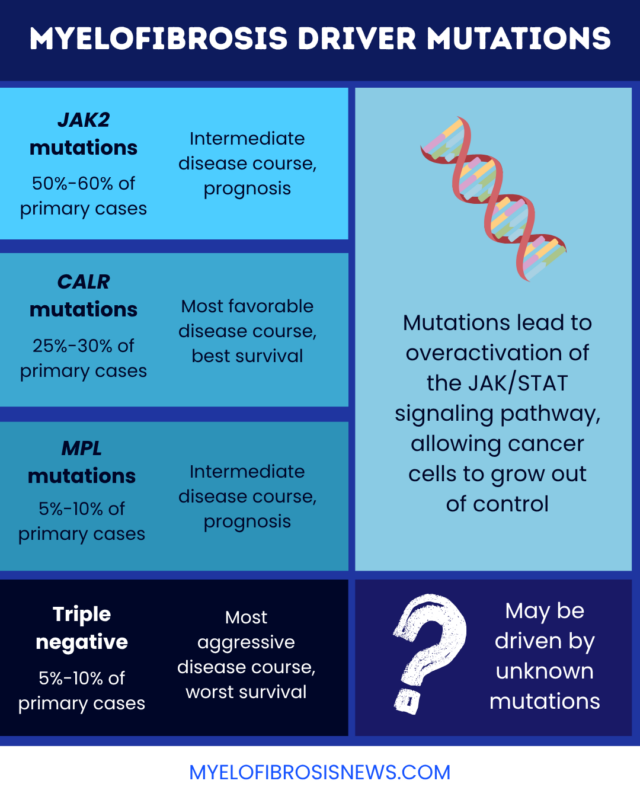
- JAK2 mutations: These account for about 50%-60% of primary myelofibrosis cases and are linked to an intermediate disease course with a higher risk of blood clots.
- CALR mutations: These mutations account for about 25%-30% of primary myelofibrosis cases and tend to have the most favorable disease course and best survival.
- MPL mutations: These account for about 5%-10% of primary myelofibrosis cases and are linked to an intermediate disease course, similar to that seen with JAK2 mutations.
- Triple-negative: This type of mutation accounts for about 5%-10% of primary myelofibrosis cases and is associated with the most aggressive disease course and poorest treatment responses.
Over time, myelofibrosis cells may acquire additional non-driver mutations in other genes that can further influence disease progression and prognosis. Some of the implicated genes are ASXL1, SRSF2, EZH2, and IDH1/2.
Risk factors
Researchers don’t know why blood stem cells become abnormal in myelofibrosis, but certain risk factors have been identified. These factors are not established myelofibrosis causes and they don’t mean a person will definitely develop the disease. They do, however, increase the chances that blood stem cells will become abnormal.
Myelofibrosis risk factors include:
- Age: Myelofibrosis is most often diagnosed in people older than 60.
- Pre-existing blood disorders: As many as about 15% of people with PV or ET, will develop myelofibrosis.
- Benzene exposure: High-level exposure to industrial chemicals, particularly benzene, can damage cellular DNA and has been linked to myelofibrosis.
- Radiation exposure: Very high exposure to ionizing radiation, such as from nuclear accidents or intensive radiation therapy, increases the risk of myelofibrosis.
Some studies also find that myelofibrosis is slightly more common in men than in women.
Although myelofibrosis is not directly inherited, a family history of MPNs is present in about 10% of cases. First-degree relatives, meaning parents, siblings and children, of people with MPNs have about a five- to sevenfold increased risk of developing a driver mutation. In such cases, there may be a genetic predisposition that makes the acquisition of driver mutations later in life more likely.
Myelofibrosis News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by