Inrebic (fedratinib) for myelofibrosis
What is Inrebic for myelofibrosis?
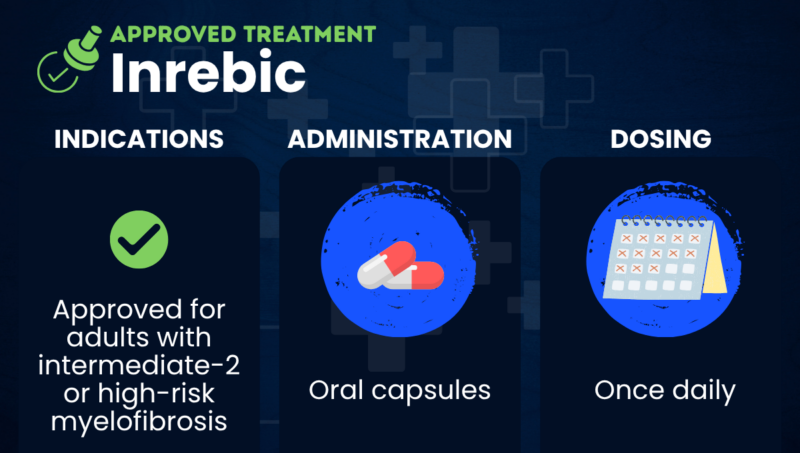
Inrebic (fedratinib) is an oral therapy approved in North America and Europe to treat certain adults with intermediate- or high-risk myelofibrosis.
In myelofibrosis, abnormal blood cells are overproduced in the bone marrow, eventually leading to scarring that further inhibits healthy blood cell production. The spleen then attempts to compensate by making some blood cells and becomes enlarged.
Inrebic belongs to a class of treatments called JAK inhibitors, which work to suppress an overactive signaling pathway that’s linked to abnormal blood cell growth in myelofibrosis. In doing so, the medication is expected to control aberrant blood cell growth, helping to shrink the spleen and ease other symptoms.
Taken in the form of oral capsules, Inrebic is marketed by Bristol Myers Squibb.
Therapy snapshot
| Brand name | Inrebic |
| Chemical name | Fedratinib |
| Usage | Used to treat certain adults with myelofibrosis |
| Administration | Oral capsules |
Who can take Inrebic?
In the U.S., Inrebic is approved for the treatment of adults with intermediate-2 or high-risk myelofibrosis, including primary myelofibrosis or myelofibrosis secondary to polycythemia vera or essential thrombocythemia. It is similarly approved in the European Union and Canada.
There are no contraindications for the use of Inrebic. However, the medication comes with a boxed warning that it may cause serious or fatal brain dysfunction, known as encephalopathy, including a neurological emergency called Wernicke’s encephalopathy. Because Wernicke’s encephalopathy is caused by low levels of thiamine (vitamin B1), Inrebic shouldn’t be used in people with a thiamine deficiency.
How is Inrebic administered?
Inrebic comes in the form of oral capsules to be taken once daily, with or without food, at a recommended dose of 400 mg for people with platelet counts of at least 50 billion per liter. Platelets are the small cell fragments that help blood clot.
Taking the medication with a high-fat meal may reduce the incidence of nausea and vomiting.
For people who have difficulty swallowing capsules whole, the capsules’ contents may be dissolved in Ensure Plus liquid nutritional supplement and administered via a feeding tube.
The recommended dose of Inrebic may have to be reduced in people using certain medications or who have severe kidney problems. It may also be adjusted if certain adverse events occur, such as low blood cell counts, gastrointestinal problems, or signs of liver toxicity.
Inrebic in clinical trials
Inrebic’s U.S. approval was largely supported by data from the Phase 3 JAKARTA trial (NCT01437787), which involved 289 adults with intermediate-2 or high-risk myelofibrosis and an enlarged spleen. Participants were randomly assigned to receive either Inrebic, at a dose of 400 or 500 mg, or a placebo, once daily, for at least six cycles.
Study results after about six months of treatment showed that, compared with the placebo, Inrebic at its now-approved 400 mg dose led to:
- a significantly higher proportion of patients who achieved at least a 35% reduction in spleen volume (37% vs. 1%)
- a significantly higher proportion of patients who achieved at least a 50% reduction in overall symptom burden (40% vs. 9%)
Inrebic side effects
The most common side effects associated with Inrebic were:
- diarrhea
- nausea
- anemia, or a lack of healthy red blood cells
- vomiting
According to the boxed warning, Inrebic increases the risk of serious and potentially fatal neurological complications, such as Wernicke’s encephalopathy, which is caused by a deficiency in thiamine.
For this reason, thiamine levels must be checked in all patients before Inrebic is started, and again throughout treatment. For people with a thiamine deficiency, Inrebic should not be started until thiamine levels are replenished. Throughout treatment with Inrebic, all patients should receive daily oral thiamine supplements and have thiamine levels checked as appropriate.
Any change in mental status, confusion, or memory impairment should raise concern for encephalopathy. If encephalopathy is suspected, Inrebic should be discontinued immediately, and thiamine infusions started, with careful monitoring until symptoms resolve and thiamine levels normalize.
Inrebic also comes with warnings for other uncommon but potentially serious side effects for which patients will be carefully monitored, including:
- anemia and low levels of platelets (thrombocytopenia)
- gastrointestinal problems, such as severe diarrhea, nausea, or vomiting
- liver toxicity
- elevated digestive enzymes, which could reflect inflammation in the pancreas
- eye inflammation
- major cardiac events
- blood clot-related complications
- secondary cancers, especially in current or past smokers
If these complications arise, they will be treated as clinically appropriate. In some cases, Inrebic may need to be paused, discontinued, or have its dose adjusted.
The symptoms of myelofibrosis could get suddenly worse when Inrebic is paused or discontinued. Should this occur, patients will be managed with supportive care and a restart of Inrebic may be considered. When possible, gradual dose tapering may help avoid symptom rebound.
People on Inrebic are advised not to breastfeed during treatment and for at least one month after the last dose.
Myelofibrosis News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by