Vonjo (pacritinib) for myelofibrosis
What is Vonjo for myelofibrosis?
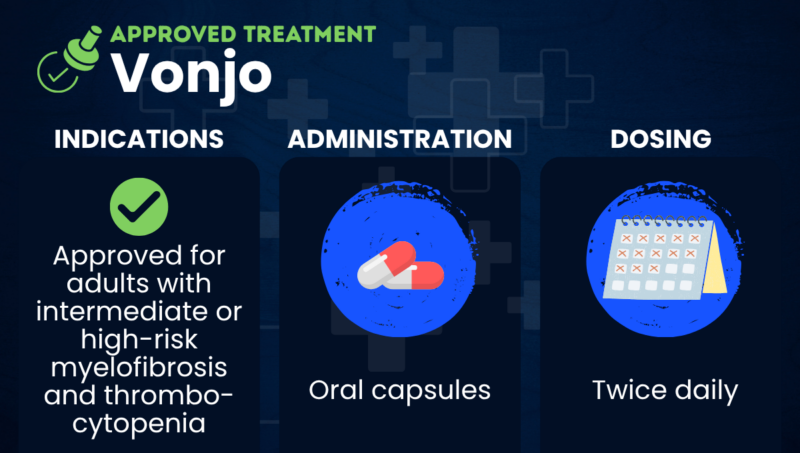
Vonjo (pacritinib) is an oral therapy that’s approved for the treatment of adults with intermediate or high-risk myelofibrosis who have severe thrombocytopenia, or low blood platelet counts.
In myelofibrosis, overactivation of the JAK/STAT signaling pathway in blood stem cells leads to the excessive production of abnormal blood cells that cause inflammation and scarring. Over time, the bone marrow’s ability to make new healthy blood cells is impaired, leading to low numbers of blood cells, including platelets, red blood cells (anemia), and white blood cells.
Vonjo is a JAK inhibitor that blocks the overactivation of JAK/STAT signaling. It is specifically designed to target JAK proteins that contribute to abnormal cell growth and inflammation in myelofibrosis, particularly JAK2. Other medications in its class also target JAK1, which can exacerbate thrombocytopenia. Vonjo also inhibits other proteins that may help decrease inflammation and combat anemia.
The therapy is available as oral capsules and is marketed by Sobi. It earned accelerated approval in the U.S. for myelofibrosis, allowing it to be marketed based on preliminary evidence that it is likely to benefit patients. Continued approval is contingent upon verification of its benefits in additional clinical studies.
Sobi is also studying Vonjo as a potential treatment for chronic myelomonocytic leukemia, another blood cancer, and a rare inflammatory condition called VEXAS.
Therapy snapshot
| Brand name | Vonjo |
| Chemical name | Pacritinib |
| Usage | Used to treat certain adults with myelofibrosis and thrombocytopenia |
| Administration | Oral capsules |
Who can take Vonjo?
In the U.S., Vonjo is conditionally approved for adults with intermediate or high-risk myelofibrosis who have a platelet count below 50 billion per liter. This includes people with primary myelofibrosis and myelofibrosis secondary to polycythemia vera or essential thrombocythemia.
Vonjo is contraindicated (should not be used by) in people taking other medications with strong effects on CYP3A4 enzymes, as they can alter exposure to Vonjo, potentially increasing the risk of side effects or impairing therapy efficacy.
Vonjo should also generally be avoided in people with severe kidney impairments.
How is Vonjo administered?
Vonjo is available as oral capsules to be taken twice daily at a recommended dose of 200 mg. The capsules can be taken with or without food. They should be swallowed whole, and never opened, broken, or chewed.
The recommended dose may have to be adjusted in people using certain medications or who have severe liver impairments. It may also be adjusted if certain side effects occur.
The use of Vonjo should be paused a week before elective surgeries or invasive procedures due to an increased risk of bleeding. It can be restarted once stable bleed control is assured.
Vonjo in clinical trials
Vonjo’s conditional U.S. approval was mainly supported by data from the Phase 3 PERSIST-2 clinical trial (NCT02055781), which involved adults with intermediate or high-risk myelofibrosis, an enlarged spleen, and thrombocytopenia, who were randomly assigned to receive Vonjo or the best available standard care for about six months.
The main efficacy analysis on the label focused on patients with initial platelet counts below 50 billion per liter who received either the now-approved regimen of Vonjo (200 mg, twice daily) or the best available standard therapy. Results showed that significantly more people on Vonjo achieved at least a 35% reduction in spleen volume compared with the best available therapy group (29% vs. 3.1%).
The ongoing Phase 3 PACIFICA trial (NCT03165734) is now seeking to confirm the clinical benefits of Vonjo compared with standard-of-care treatments in people with myelofibrosis and severe thrombocytopenia. Its main goals are to evaluate changes in spleen volume and total symptom burden after about six months.
Vonjo side effects
The most common side effects of Vonjo include:
- diarrhea
- thrombocytopenia
- nausea
- anemia
- swelling in the legs, ankles, feet, or hands
Vonjo’s label also comes with warnings for potentially serious adverse events, including:
- serious or fatal bleeding complications
- significant diarrhea
- thrombocytopenia
- heart rhythm abnormalities or major cardiac events
- blood clot-related complications
- secondary cancers, particularly in former or current smokers
- increased risk of infections
Patients will be carefully monitored for these complications and treated appropriately if they arise, which may involve pausing Vonjo, adjusting the dose, or discontinuing treatment entirely. Vonjo shouldn’t be started in people with active bleeds or infections.
The symptoms of myelofibrosis could get worse if Vonjo is discontinued suddenly, and the medication should be gradually tapered when it is safe to do so. If symptoms reemerge, patients will be treated with supportive care, and reinitiation of Vonjo may be discussed.
Vonjo may reduce the effectiveness of most hormonal contraceptives, except for intrauterine devices containing levonorgestrel. If contraception is needed or desired, individuals should discuss with their doctor an alternative method of contraception while using Vonjo and for 30 days after the last dose.
Patients are also advised not to breastfeed while using the medication and for two weeks after the last dose.
Myelofibrosis News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by