Ojjaara (momelotinib) for myelofibrosis
What is Ojjaara for myelofibrosis?
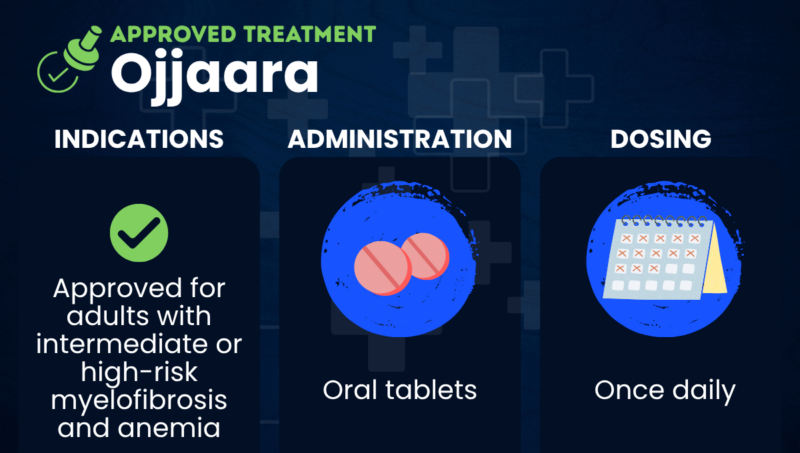
Ojjaara (momelotinib) is an approved oral therapy used to treat adults with intermediate or high-risk myelofibrosis who have anemia, or a lack of healthy red blood cells.
Myelofibrosis is a rare blood cancer in which abnormal blood cells are produced in the bone marrow, leading to scarring and inflammation that impair normal bone marrow function. This abnormal cell production is linked to overactivation of the JAK/STAT signaling pathway.
As a JAK inhibitor, Ojjaara works to put the brakes on that signaling pathway. This helps reduce aberrant cell production, in turn reducing the size of the spleen, and easing other myelofibrosis symptoms.
While it works similarly to other JAK inhibitors, Ojjaara is the only one specifically indicated for people who have anemia. That is because it also inhibits the activity of a liver protein called activin A receptor type 1, which helps increase red blood cell counts.
The therapy is marketed by GlaxoSmithKline and is taken as oral tablets. In certain areas, including the European Union, it is sold under the brand name Omjjara.
Therapy snapshot
| Brand name | Ojjaara |
| Chemical name | Momelotinib |
| Usage | Used to treat certain adults with myelofibrosis and anemia |
| Administration | Oral tablets |
Who can take Ojjaara?
Ojjaara is approved in the U.S. for the treatment of adults with intermediate or high-risk myelofibrosis — including primary myelofibrosis and myelofibrosis secondary to polycythemia vera or essential thrombocythemia — who also have anemia.
It holds similar approvals in Canada and the European Union.
There are no contraindications for Ojjaara’s use.
How is Ojjaara administered?
Ojjaara is available as oral tablets to be swallowed whole once daily, with or without food. The tablets should not be cut, crushed, or chewed. The recommended dose is 200 mg, which may be adjusted in people with severe liver problems or who are using certain medications.
Patients will have blood tests throughout treatment to monitor for liver toxicity or low blood cell counts. If these or other side effects occur, the Ojjaara dose may need to be adjusted or treatment paused.
Ojjaara in clinical trials
Ojjaara’s approval for the treatment of adults with intermediate and high-risk myelofibrosis was largely supported by data from two Phase 3 studies: MOMENTUM (NCT04173494) and SIMPLIFY-1 (NCT01969838).
- MOMENTUM involved 195 symptomatic and anemic adults with myelofibrosis who had previously received an approved JAK inhibitor therapy. Participants received either Ojjaara or danazol, a steroid medication used to manage anemia, for six months. A significantly greater proportion of people in the Ojjaara group experienced at least a 50% reduction in symptom burden compared with the danazol group (25% vs. 9%). Also, significantly more people on Ojjaara stopped needing blood transfusions for anemia and had a meaningful reduction in spleen size.
- SIMPLIFY involved adults with myelofibrosis who had never received a JAK inhibitor, and who received either Ojjaara or the approved JAK inhibitor Jakafi (ruxolitinib) for six months. The approval was mainly supported by data from a subset of 181 participants with anemia, which showed that a similar proportion of participants in either group achieved at least a 35% reduction in spleen volume.
Ojjaara side effects
The most common side effects of Ojjaara include:
- low platelet counts (thrombocytopenia)
- bleeding
- bacterial infections
- fatigue
- dizziness
- diarrhea
- nausea
Ojjaara also comes with warnings that it could cause potentially serious side effects, for which patients must be carefully monitored. These include:
- increased risk of serious or fatal infections, including reactivation of the hepatitis B virus
- thrombocytopenia or low levels of neutrophils (neutropenia), a type of infection-fighting immune cells
- liver toxicity
- severe skin reactions
- major cardiovascular events
- blood clot-related complications
- secondary cancers, especially in current or former smokers
If these side effects occur, they will be managed as appropriate. In some cases, Ojjaara may need to be paused, discontinued, or its dose adjusted.
The symptoms of Ojjaara may get worse when treatment is paused or discontinued. If this occurs, patients will receive supportive care, and reinitiation of Ojjaara may be considered. If treatment is being paused for reasons other than life-threatening toxicities, the dose may be gradually tapered rather than stopped abruptly.
Ojjaara could cause harm to a developing fetus. Female patients of reproductive potential are advised to use highly effective contraception during treatment and for at least a week after the last dose. Female patients are also advised not to breastfeed while using the medication and for at least a week after the last dose.
Myelofibrosis News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by