Jakafi (ruxolitinib) for myelofibrosis
What is Jakafi for myelofibrosis?
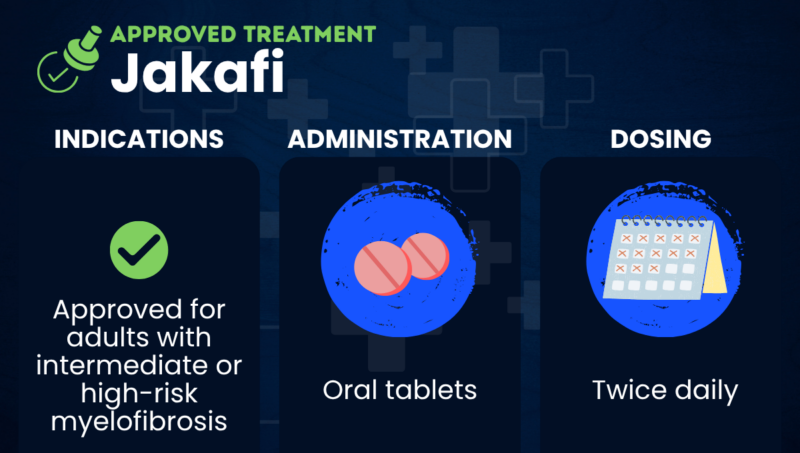
Jakafi (ruxolitinib) is an oral therapy approved for the treatment of certain adults with intermediate or high-risk myelofibrosis.
Myelofibrosis is a blood cancer in which abnormal blood cell production leads to inflammation and scarring in the bone marrow. It is generally associated with overaction of the JAK-STAT signaling pathway that’s involved in blood cell production.
Jakafi belongs to a class of therapies called JAK inhibitors, which work to dampen this overactive signaling pathway. This helps reduce abnormal cell growth and ease related symptoms, such as an enlarged spleen, fatigue, and night sweats.
Taken in the form of oral tablets, Jakafi is marketed in the U.S. by Incyte, and elsewhere by Novartis under the brand name Jakavi. It is also approved for treating certain people with the blood disorder polycythemia vera (PV) and the post-transplant complication graft-versus-host disease.
Therapy snapshot
| Brand name | Jakafi |
| Chemical name | Ruxolitinib |
| Usage | Used to treat certain adults with myelofibrosis |
| Administration | Oral tablets |
Who with myelofibrosis can take Jakafi?
In the U.S., Jakafi is approved for adults with intermediate or high-risk myelofibrosis, including primary myelofibrosis, post-PV myelofibrosis, and post-essential thrombocythemia myelofibrosis.
The medication is also approved in the European Union and Canada for treating disease-related symptoms in adults with myelofibrosis.
There are no contraindications for Jakafi’s use.
How is Jakafi administered in myelofibrosis?
Jakafi is administered as oral tablets, taken twice daily, with or without food. For people unable to swallow, the tablets can be suspended in about 40 mL of water and administered through a feeding tube.
The recommended starting dose is based on a person’s blood levels of platelets, the cell fragments involved in blood clotting:
- greater than 200 billion per liter: 20 mg twice daily
- 100 billion to 200 billion per liter: 15 mg twice daily
- 50 billion to 99 billion per liter: 5 mg twice daily
The dose is then individualized based on safety and efficacy. It can be increased as needed for efficacy to 10 mg twice daily for people with platelet counts under 100 billion per liter and to 25 mg twice daily for people with higher platelet levels.
Blood counts will be monitored regularly during the dose adjustment phase, and thereafter as clinically indicated. If blood cell counts get too low, treatment may need to be modified or interrupted. When restarting treatment after a safety pause, a different dosing regimen may be required.
Jakafi doses may also need to be adjusted in patients who are taking other medications, and in those who have moderate-to-severe kidney impairments or kidney failure.
Jakafi in myelofibrosis clinical trials
Jakafi’s approval for myelofibrosis was largely supported by two Phase 3 clinical trials, both involving adults with intermediate or high-risk myelofibrosis and an enlarged spleen:
- The COMFORT-1 trial (NCT00952289) enrolled 309 patients who did not respond sufficiently to or were not candidates for available treatments. They were randomly assigned to receive either Jakafi or a placebo. Results showed that 41.9% of people given Jakafi achieved at least a 35% reduction in spleen size after six months, compared with less than 1% of those on placebo. Overall symptom burden was also reduced with the treatment.
- The COMFORT-2 trial (NCT00934544) was an open-label study involving 219 patients, who received either Jakafi or the best available therapy. Results showed that nearly 30% of Jakafi-treated patients achieved at least a 35% reduction in spleen size after nearly a year, compared with none in the standard care group.
Additional analyses demonstrated survival benefits with Jakafi relative to the comparator group in both studies.
Jakafi side effects
In people with myelofibrosis, the most common side effects of Jakafi include:
- thrombocytopenia
- anemia, or a lack of healthy red blood cells
- bruising
- dizziness
- headache
- diarrhea
Jakafi also comes with warnings for potential serious side effects, for which patients will require careful monitoring, including:
- low blood cell counts
- increased risk of serious infections, including tuberculosis, herpes, the rare brain infection progressive multifocal leukoencephalopathy, and reactivation of the hepatitis B virus
- increased risk of non-melanoma skin cancer
- high levels of fats in the blood, including cholesterol
- major cardiovascular events, including a heart attack or stroke
- blood clot-related complications
- secondary cancers, especially in current or former smokers
If any of these occur, they will be treated as appropriate. In some cases, the Jakafi dose may need to be reduced or treatment interrupted. Jakafi shouldn’t be initiated in people with active serious infections.
Myelofibrosis symptoms could get suddenly worse when treatment is interrupted or discontinued. If that occurs, patients will receive supportive care, and reinitiation of Jakafi may be considered. When stopping Jakafi for reasons other than low blood cell counts, the dose should be gradually tapered.
Patients are also advised not to breastfeed while using the medication and for two weeks after the last dose.
Myelofibrosis News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by