Navtemadlin for myelofibrosis
What is navtemadlin for myelofibrosis?
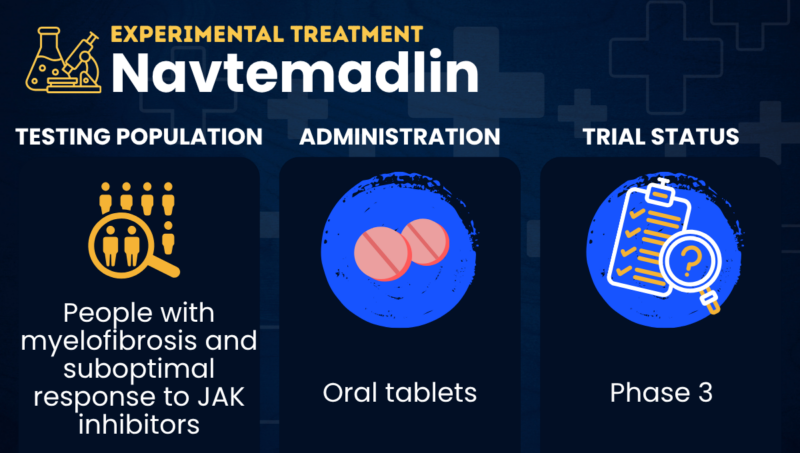
Navtemadlin (KRT-232) is an experimental oral treatment that’s being tested as a possible disease-modifying therapy for people with myelofibrosis who did not respond well enough to JAK inhibitors.
Abnormal, cancerous cells grow out of control in the bone marrow of people with myelofibrosis, causing inflammation and scarring that inhibits healthy blood cell production.
Navtemadlin is designed to control this abnormal cell growth by boosting the activity of the tumor-suppressing protein p53. Normally, p53 isn’t very active, but it becomes activated in response to stress signals, such as cellular damage or cancer-related signaling, to help kill off the problematic cells.
A protein called MDM2 regulates p53. In healthy cells, activated MDM2 keeps p53 levels low. Cancer cells sometimes abnormally elevate MDM2 production to allow their uncontrolled growth. Navtemadlin blocks MDM2, allowing p53 to be activated. This is expected to help control cancer cell growth and slow disease progression.
Kartos Therapeutics is developing Navtemadlin. Several clinical trials have tested navtemadlin in people with myelofibrosis, including Phase 3 studies evaluating the therapy as a standalone treatment or as an add-on to the approved JAK inhibitor Jakafi (ruxolitinib).
The medication has received fast-track designation in the U.S. for the treatment of certain people with myelofibrosis. This status is designed to expedite the therapy’s development and regulatory review.
Therapy snapshot
| Treatment name | Navtemadlin |
| Administration | Oral tablets |
| Clinical testing | Currently in Phase 3 testing |
How will navtemadlin be administered in myelofibrosis?
Navtemadlin comes in the form of oral tablets. In a Phase 3 trial that tested navtemadlin as a standalone therapy, the treatment was administered once daily for the first week of repeating four-week treatment cycles, at a dose of 240 mg.
In another Phase 3 trial testing navtemadlin in combination with Jakafi, the treatment is being given on a similar schedule.
Navtemadlin in myelofibrosis clinical trials
A few clinical trials have tested navtemadlin in people with myelofibrosis:
- The Phase 3 BOREAS trial (NCT03662126) compared navtemadlin with best available standard therapies among people with myelofibrosis who were no longer benefiting from JAK inhibitors. The data showed that more people on navtemadlin achieved at least a 35% reduction in spleen volume (15% vs. 5%) and at least a 50% reduction in total symptom burden (24% vs. 12%) compared with those on standard treatment. The therapy also showed signs of reducing bone marrow fibrosis.
- The Phase 1/2 KRT-232-109 trial (NCT04485260) evaluated the safety and efficacy of navtemadlin in combination with Jakafi in people with myelofibrosis who had a suboptimal response to Jakafi. Data showed the addition of navtemadlin was associated with clinically meaningful improvements in measures of spleen volume and symptom burden.
The ongoing Phase 3 POIESIS trial (NCT06479135) is now further evaluating the potential benefits of navtemadlin as an add-on to Jakafi in people who had a suboptimal response to Jakafi during the trial’s run-in period. The study primarily aims to assess the effects of treatment on spleen size and total symptom reduction after about six months. Top-line data are expected in 2026.
Navtemadlin side effects
In the BOREAS trial, the most common side effects occurring in people given navtemadlin included:
- low blood cell counts
- nausea or vomiting
- diarrhea or constipation
- decreased appetite
- fatigue or general weakness
- swelling
- upper abdominal pain
Myelofibrosis News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by