Pelabresib for myelofibrosis
What is pelabresib for myelofibrosis?
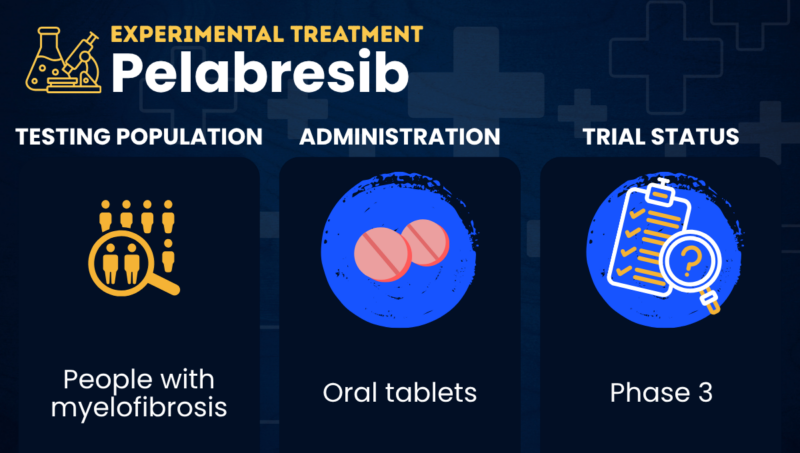
Pelabresib is an experimental oral therapy that’s being tested as a possible treatment for myelofibrosis when used alongside the approved JAK inhibitor Jakafi (ruxolitinib).
Myelofibrosis is a rare blood cancer in which an excess of abnormal blood cells is produced in the bone marrow. These cancerous cells release inflammatory substances that contribute to bone marrow scarring (fibrosis), inhibiting the production of healthy blood cells.
Pelabresib is designed to dampen pro-inflammatory and fibrosis-promoting signaling by inhibiting proteins in the bromodomain and extraterminal domain (BET) family. Preclinical studies show that BET inhibition reduced spleen size, bone marrow scarring, and inflammation in a mouse model of myelofibrosis.
JAK inhibitors, the current standard of care for myelofibrosis, help to ease symptoms but have a limited ability to slow disease progression. It’s thought that BET inhibitors may complement JAK therapy to better target disease processes in the bone marrow and lead to deeper treatment responses.
Pelabresib is being developed by Novartis, which also sells Jakafi in markets outside the U.S. under the brand name Jakavi. The company has plans to seek approval of pelabresib for myelofibrosis in the European Union in 2026. Regulatory applications in the U.S., China, and Japan are pending data from an additional Phase 3 study.
U.S. regulators have granted pelabresib orphan drug and fast-track designations for the treatment of myelofibrosis. These statuses are intended to speed its development and regulatory review.
Therapy snapshot
| Treatment name | Pelabresib |
| Administration | Oral tablets |
| Clinical testing | Currently in Phase 3 testing |
How will pelabresib be administered in myelofibrosis?
In Phase 3 clinical trials, people with myelofibrosis are receiving pelabresib in the form of oral tablets, taken in repeating three-week cycles.
During each cycle, pelabresib (125 mg) is taken once daily for the first two weeks, followed by a one-week break. It is given along with Jakafi, which is taken twice daily without breaks.
Pelabresib in myelofibrosis clinical trials
Two main clinical trials have tested the combination of pelabresib and Jakafi in adults with myelofibrosis: the open-label Phase 2 MANIFEST study (NCT02158858) and the placebo-controlled Phase 3 MANFEST-2 study (NCT04603495 ). Both studies involved people who had never received a JAK inhibitor.
- Data from MANIFEST showed that 68% of participants who received the treatment combination achieved at least a 35% reduction in spleen size after six months, and more than half (56%) achieved at least a 50% reduction in total symptom burden.
- Data from MANIFEST-2 showed that significantly more people on pelabresib and Jakafi than on Jakafi alone experienced a 35% reduction in spleen size after six months (65.9% vs. 35.2%). More people in the combination group experienced at least a 50% reduction in symptom burden (52.3% vs. 46.3%), but the difference wasn’t statistically significant. Long-term trial data showed that the benefits of the combination therapy were sustained or further improved with up to two years of treatment.
A Phase 3 trial called MANIFEST-3 (NCT07357727) will further evaluate the clinical benefits and safety of pelabresib and Jakafi compared with Jakafi alone among people with myelofibrosis. The study is expected to conclude in 2030.
Pelabresib side effects
In the MANIFEST-2 trial, the most common side effects reported in people treated with pelabresib included:
- a lack of healthy red blood cells (anemia)
- low levels of platelets, the cell fragments that help blood clot
- altered taste
- diarrhea or constipation
- nausea
- elevated liver enzymes
- fatigue
Myelofibrosis News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by