Selinexor for myelofibrosis
What is selinexor for myelofibrosis?
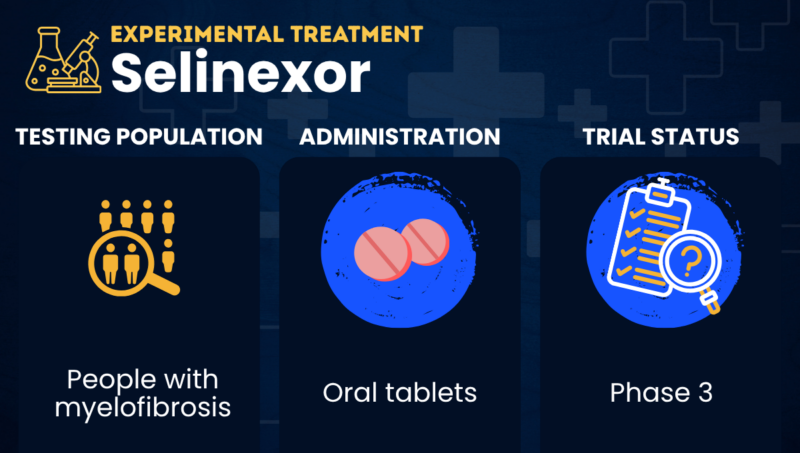
Selinexor is an investigational oral therapy that’s being tested as a possible treatment for myelofibrosis when used either alone or in combination with the approved JAK inhibitor Jakafi (ruxolitinib).
In myelofibrosis, abnormal blood cells are overproduced in the bone marrow. The cancerous cells release inflammatory molecules that cause scarring (fibrosis) in the marrow, which inhibits healthy blood cell production. The spleen, which tries to help out by producing blood cells, becomes enlarged.
Selinexor works by inhibiting exportin-1 (XPO1), a protein involved in shuttling regulatory proteins out of a cell’s nucleus, where its DNA is stored. Among the proteins it normally moves are those involved in tumor suppression and growth regulation. By blocking XPO1, selinexor causes these proteins to accumulate in the nucleus, where they suppress signaling pathways that cancerous cells use to grow and survive.
Scientists believe that the combination of selinexor with Jakafi, which works to slow abnormal cancer cell growth by a different mechanism, will lead to enhanced therapeutic outcomes.
Karyopharm Therapeutics is developing Selinexor. It is already approved in the U.S. under the brand name Xpovio for the treatment of multiple myeloma and certain types of diffuse large B-cell lymphoma. Karyopharm plans to meet with U.S. regulators to discuss submitting an application seeking approval of selinexor for myelofibrosis.
The medication also holds fast-track and orphan drug designations in the U.S. for the treatment of myelofibrosis. These statuses are intended to speed its development and regulatory review.
Therapy snapshot
| Treatment name | Selinexor |
| Administration | Oral tablets |
| Clinical testing | Currently in Phase 3 testing |
How will selinexor be administered in myelofibrosis?
Selinexor comes in the form of oral tablets. In a Phase 3 trial involving people with myelofibrosis, selinexor was administered at a weekly dose of 60 mg, in combination with twice-daily Jakafi.
When given as a single agent (monotherapy) in a Phase 2 trial, selinexor was administered at a weekly dose of 80 mg for the first two months, then reduced to 60 mg weekly.
Selinexor in myelofibrosis clinical trials
A few clinical trials have tested selinexor in people with myelofibrosis:
- The Phase 3 SENTRY trial (NCT04562389) tested the combination of selinexor and Jakafi as a first-line treatment for adults with myelofibrosis. Top-line results showed that significantly more people on the combination therapy achieved a 35% reduction in spleen size after six months compared with those on Jakafi alone (50% vs. 28%), while the reduction in total symptom burden was similar. The risk of death was reduced by 57% in the combination group compared with the Jakafi-only group.
- The Phase 2 XPORT-MF-035 trial (NCT04562870) evaluated the benefits of selinexor monotherapy among people with heavily pretreated myelofibrosis. The results showed that compared with a physician’s choice of standard treatments, selinexor was associated with higher rates of spleen volume reduction, reduced need for blood transfusions, and greater reductions in inflammatory molecules known to contribute to the disease.
An ongoing Phase 2 clinical trial called SENTRY-2 (NCT05980806) is evaluating the efficacy of selinexor in myelofibrosis patients who have not received a JAK inhibitor, including those with normal levels of platelets or mild-to-moderately low platelet counts (thrombocytopenia). Top-line data are expected this year.
Selinexor side effects
In the SENTRY trial, the most common treatment-emergent side effects observed in people who received selinexor in combination with Jakafi included:
- low blood cell counts
- nausea
- constipation
In the XPORT-MF-035 trial, the most common treatment-emergent side effects seen in people who received selinexor alone included:
- decreased weight
- low blood cell counts
- general weakness
- nausea
- upper respiratory tract infection
Myelofibrosis News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by